Gravimetric analysis is a method of quantitative chemical analysis in which the mass of a compound is used to determine its quantity. This technique is particularly useful for determining the concentration of a soluble chloride, such as sodium chloride (common table salt). In this essay, we will discuss the general principles of gravimetric analysis and the specific steps involved in performing a gravimetric analysis of a soluble chloride.
The basic principle behind gravimetric analysis is the measurement of mass. In order to determine the mass of a compound, it must first be isolated from the rest of the sample. This is typically done through a process called precipitation, in which the compound is transformed into a solid that can be easily separated and weighed.
The specific steps involved in a gravimetric analysis of a soluble chloride depend on the particular chloride being analyzed and the desired end result. However, there are some general steps that are followed in most gravimetric analyses.
First, the sample is prepared by dissolving it in a suitable solvent. The solvent should be chosen based on the solubility of the compound being analyzed and the desired end result. For example, water may be used as a solvent for a soluble chloride if the goal is to determine the mass of the chloride.
Next, the precipitating reagent is added to the sample. This reagent is chosen based on the solubility of the compound being analyzed and the desired end result. For example, a soluble chloride may be precipitated as a silver chloride by adding a silver nitrate solution to the sample.
Once the precipitate has formed, it is allowed to settle to the bottom of the container. The supernatant liquid is then carefully decanted, leaving the precipitate behind. The precipitate is then washed with a solvent to remove any impurities that may have been present in the sample.
Finally, the precipitate is dried and weighed to determine its mass. This mass can then be used to calculate the concentration of the soluble chloride in the original sample.
In summary, gravimetric analysis is a powerful tool for determining the concentration of a soluble chloride. By following the steps outlined above, it is possible to accurately and precisely determine the mass of a compound, which can be used to calculate its concentration.
Essay writing is a common form of academic writing that is used to assess a student's understanding of a particular subject or topic. There are several different types of essays that students may be asked to write, and each type serves a specific purpose. Some common types of essays include narrative essays, descriptive essays, expository essays, persuasive essays, and compare and contrast essays.
Narrative essays are stories that are written in the first person and typically include a plot, characters, and a theme. They are often used to describe personal experiences or events, and the goal is to engage the reader by telling a compelling story.
Descriptive essays are written to describe a person, place, or thing in detail. The goal is to use vivid language to paint a picture in the reader's mind and help them feel as if they are experiencing the thing being described.
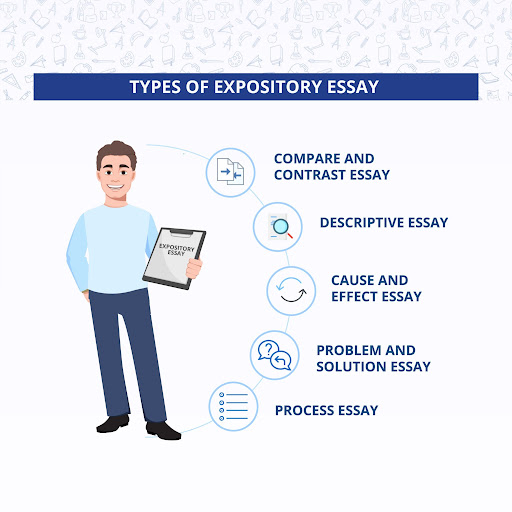
Expository essays are written to explain a topic or concept. The goal is to provide the reader with information and clarify any misunderstandings or confusion about the topic.
Persuasive essays are written to convince the reader to take a specific action or adopt a particular point of view. The writer presents their argument and uses evidence and logic to try to persuade the reader to agree.
Compare and contrast essays are written to compare and contrast two or more subjects. The writer looks at the similarities and differences between the subjects and uses this information to make a point or argue a case.
In summary, there are several different types of essays that students may be asked to write, each serving a specific purpose. Understanding the different types of essays and their purposes can help students write more effective and targeted essays that meet the requirements of their assignments.