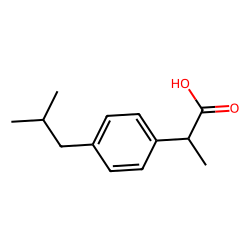
Ibuprofen is a common over-the-counter medication used to relieve pain, reduce inflammation, and reduce fever. It is a member of the nonsteroidal anti-inflammatory drug (NSAID) class of medications, which also includes other well-known drugs such as aspirin and naproxen.
One physical property of a substance is its boiling point, which is the temperature at which the substance transitions from a liquid to a gas. The boiling point of a substance is dependent on several factors, including the strength of intermolecular forces within the substance and the atmospheric pressure.
The boiling point of ibuprofen is 356°F (180°C). This is relatively high compared to the boiling points of other common substances, such as water (212°F or 100°C) and ethanol (173°F or 78°C). The high boiling point of ibuprofen is due to the presence of strong intermolecular forces within the molecules of the substance.
In general, substances with high boiling points have stronger intermolecular forces and are more difficult to vaporize. This means that it requires more energy to turn ibuprofen into a gas, and it will remain a liquid at relatively high temperatures.
It is important to note that the boiling point of ibuprofen may vary slightly depending on the specific conditions of the measurement, such as the atmospheric pressure. However, the boiling point of 356°F (180°C) is considered to be the standard boiling point for ibuprofen under standard conditions.
In conclusion, ibuprofen has a boiling point of 356°F (180°C), which is relatively high compared to other common substances. This high boiling point is due to the presence of strong intermolecular forces within the molecules of ibuprofen.