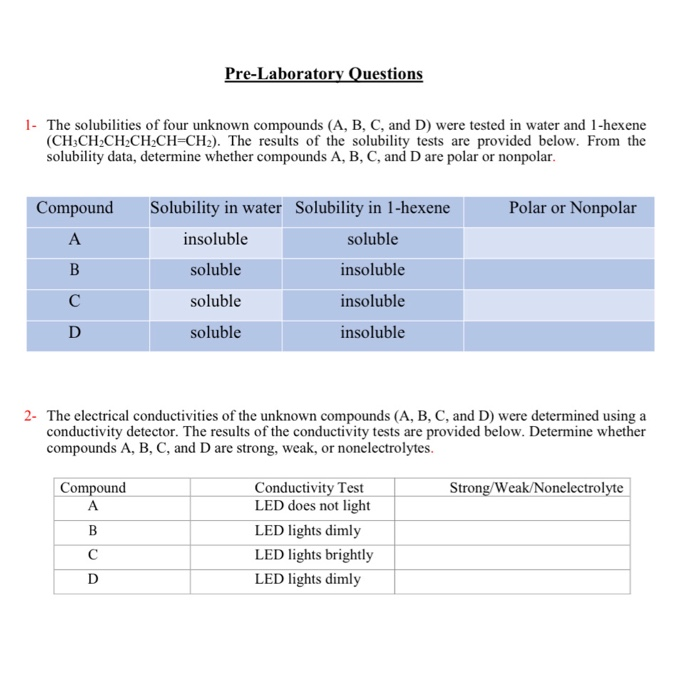
Solubility is the ability of a substance to dissolve in a solvent to form a homogeneous solution. It is an important property that can be used to identify unknown substances, as different substances have different solubilities in various solvents.
There are several ways in which solubility can be used to identify an unknown substance. One method is by performing a solubility test, in which a small amount of the unknown substance is added to a variety of solvents and the resulting solutions are observed. If the substance dissolves in a solvent, it is considered soluble in that solvent. If the substance does not dissolve, it is considered insoluble. By comparing the solubility of the unknown substance to known substances, it may be possible to determine the identity of the unknown substance.
Another method is by using solubility rules, which are guidelines that can be used to predict the solubility of a substance in a given solvent. For example, it is generally known that most ionic compounds are soluble in water, while most covalent compounds are not. By applying these rules to the unknown substance, it may be possible to determine its identity.
In addition to solubility tests and solubility rules, other techniques can be used in combination with solubility to identify an unknown substance. These include chemical reactions, spectroscopy, and microscopic examination.
For example, if the unknown substance is a metal, it may be possible to identify it by performing a flame test, in which the substance is heated in a flame and the resulting color is observed. Different metals produce different colors when heated in a flame, so this can be used as a way to identify the unknown substance.
Overall, solubility is a useful property that can be used to identify an unknown substance. By performing solubility tests, using solubility rules, and employing other techniques, it is possible to determine the identity of a substance with a high degree of accuracy.