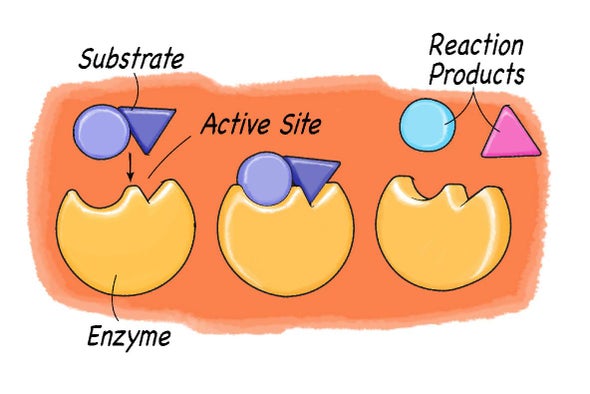
Hydrogen peroxide (H2O2) is a chemical compound that is commonly used as a disinfectant and bleach due to its ability to kill bacteria and break down organic matter. While it is a useful product, it is also potentially dangerous as it can cause harm to living tissues if it comes into contact with them in high concentrations. This is because hydrogen peroxide decomposes into oxygen and water when it comes into contact with certain enzymes, and this release of oxygen can cause damage to cells and tissues.
The enzyme that is responsible for breaking down hydrogen peroxide into oxygen and water is called catalase. This enzyme is found in almost all living cells, and it plays a crucial role in protecting the cell from the toxic effects of hydrogen peroxide. When hydrogen peroxide comes into contact with catalase, it is quickly broken down into water and oxygen, with the oxygen being released as a gas.
Catalase is a large enzyme that is made up of four subunits, each of which contains a heme group. The heme group is responsible for the enzyme's catalytic activity, and it works by transferring an electron from the hydrogen peroxide molecule to the oxygen atom, causing the oxygen atom to be released as a gas. This process is known as reduction, and it occurs very rapidly, with catalase being able to break down thousands of molecules of hydrogen peroxide per second.
While catalase is found in almost all living cells, it is particularly abundant in the liver and in red blood cells. This is because these cells are exposed to high levels of hydrogen peroxide on a regular basis, and they need a way to protect themselves from its toxic effects. In the liver, catalase helps to break down hydrogen peroxide that is produced as a byproduct of various metabolic processes, while in red blood cells, it helps to protect the cells from the oxidative stress that occurs during oxygen transport.
In conclusion, the enzyme catalase is responsible for breaking down hydrogen peroxide into oxygen and water. This enzyme is found in almost all living cells and plays a crucial role in protecting cells from the toxic effects of hydrogen peroxide. It does this by

:max_bytes(150000):strip_icc()/GettyImages-583679458-7ea6c76f50554eefb9a04054658237fa.jpg)