Water is a substance that is essential to life as we know it. It has a number of unique properties that make it particularly well suited for supporting life on Earth. In this essay, we will explore some of these properties and how they contribute to the possibility of life on our planet.
One of the most important properties of water is its ability to exist in all three states of matter (solid, liquid, and gas) at temperatures that are commonly found on Earth. This is due to the fact that water has a very high specific heat capacity, which means that it can absorb or release large amounts of heat without changing temperature significantly. This allows water to act as a natural thermoregulator, helping to stabilize temperatures and create suitable environments for living organisms.
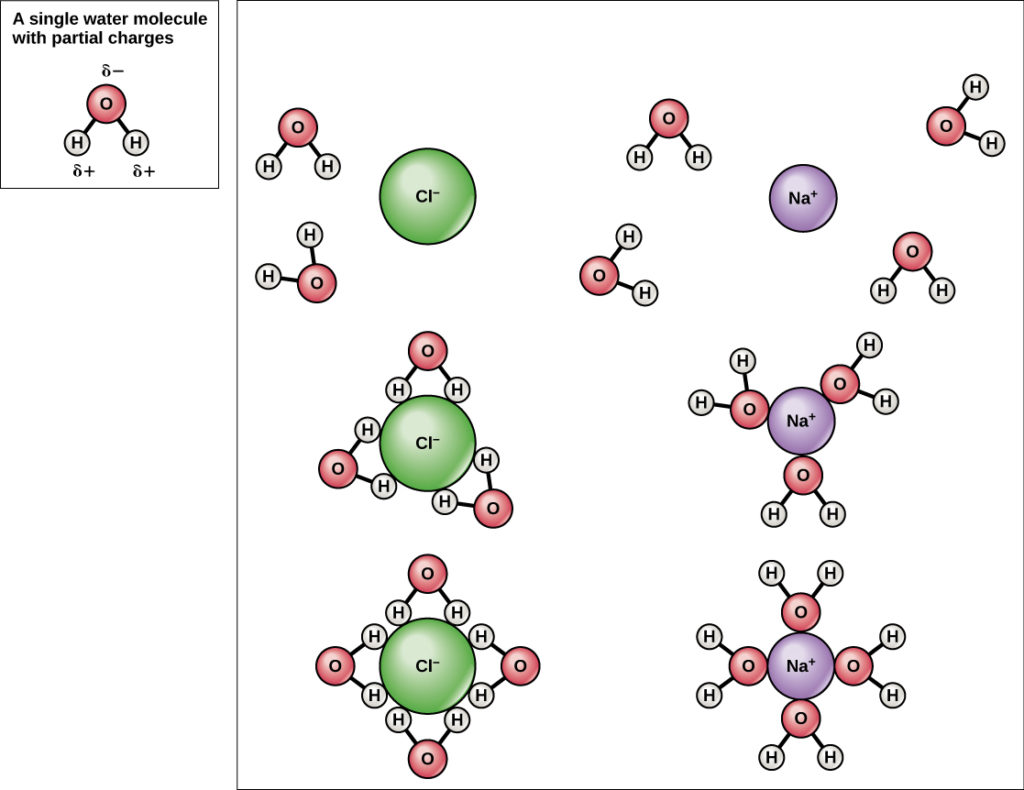
Water is also an excellent solvent, meaning that it can dissolve a wide range of substances. This is important for life because it allows the body to transport nutrients and other essential molecules throughout the body. In addition, water's ability to dissolve substances makes it possible for chemical reactions to occur, which are necessary for processes such as digestion and metabolism.
Another important property of water is its surface tension. This is the result of the attractive forces between water molecules, which cause the surface of the water to behave like an elastic membrane. This property is exploited by many living organisms, such as water striders, which are able to walk on water due to the surface tension. In addition, surface tension is important in the formation of raindrops and the ability of plants to transport water from their roots to their leaves.
Finally, water has a high heat of vaporization, which means that it requires a large amount of heat to convert it from a liquid to a gas. This property helps to regulate the temperature of the Earth by absorbing heat as it evaporates and releasing it as it condenses. It also plays a role in the water cycle, which is essential for the survival of many living organisms.
In conclusion, water has a number of unique properties that make it essential for life on Earth. Its ability to exist in all three states of matter, its effectiveness as a solvent, its surface tension, and its high heat of vaporization all contribute to its importance for sustaining life on our planet.
1 the unique properties characteristics of water make... Free Essays

This is due to water's high specific heat capacity. Each of the two hydrogen atoms shares its single electron with the oxygen atom in its outer covering to fill the two empty spaces, thus completing the shell with 8 electrons while forming two covalent bonds. One part of the molecule, the oxygen, is partially negative, and the hydrogen portion is partially positive. Additionally, water is unique in that it's less dense in solid form than as a liquid. Water has a number of unique properties that not only make it essential to survival, but also make life possible in the first place. Finally, they also share the ability to pass down their genetic information.
How do the unique properties of water support life on Earth?

This is true for naturally occurring saltwater, such as what is found in oceans, gulfs and other bodies of water, as well as manmade saltwater. The fact that water molecules naturally want to cling to one another means that water has cohesion. All of the water in the aquifer should be treated with a chemical to purify it, but no changes should be made to the injection well. The response indicates that the slightly negatively charged O atom in one water molecule is attracted to one of the slightly positively charged H atoms in another water molecule, so that a weak hydrogen bond forms between the two water molecules. . You can notice at the end of winter, there will often be a sheet of ice left on top of the melted water.
6 Features That Make Water a Unique Compound
Water in streams, rivers, and oceans distributes these materials worldwide, making them available for life. Select three of the following, and for each of the three that you have chosen, explain how the process of recognition occurs and give an example. This allows for a message to travel down a nerve. Imagining life without water is impossible, literally. It has the highest specific heat of everyday substances. Category 1: Phospholipids is located in the cell membrane as protector. These can indicate the type of structure and predict its physical properties.
What Are the Unique Properties of Water?

Viscosity The more viscosity a fluid has, the more it resists flow. Thus, it suggests that water is found in all three states, thereby fulfilling the sixth property of the compound. Which statement best helps explain the formation of the hydrogen bond represented in the figure? Addicted to online creative writing, she puts some of what she feels inside her stormy heart on paper. In fact, NASA scientists have found evidence that Mars did have quite a bit of water in the past. If ice were more dense than liquid water it would sink, and every body of water, including the oceans, would eventually freeze over and become solid ice.








