The specific heat capacity of a substance is a measure of the amount of energy required to raise the temperature of a given mass of that substance by a certain degree. It is an important property of a substance because it determines how quickly or slowly the substance will absorb or release heat when it is subjected to a temperature change. Propanol, also known as propyl alcohol or isopropyl alcohol, is a common solvent and is widely used in a variety of applications. In this essay, we will discuss the specific heat capacity of propanol and how it relates to the substance's properties and uses.
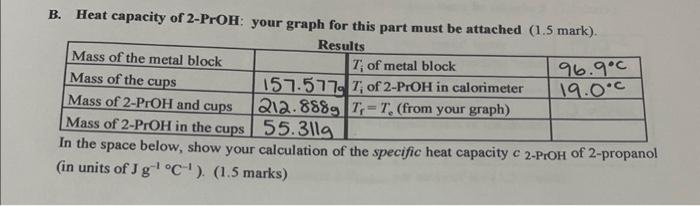
The specific heat capacity of propanol is 2.44 J/g·K. This means that it takes 2.44 joules of energy to raise the temperature of 1 gram of propanol by 1 degree Kelvin. Propanol has a relatively low specific heat capacity compared to other substances, which means that it absorbs and releases heat relatively quickly. This property makes it an effective solvent for dissolving a wide range of substances, as it can easily transfer heat to or from the dissolved material.
One of the primary uses of propanol is as a solvent for cleaning and degreasing surfaces. It is commonly used to remove dirt, oil, and other contaminants from a variety of surfaces, including metals, plastics, and glass. Propanol's low specific heat capacity makes it well suited for this purpose, as it can quickly dissolve and remove contaminants without significantly affecting the temperature of the surface being cleaned.
In addition to its use as a solvent, propanol is also used as a component of antifreeze and as a fuel additive. Its low specific heat capacity makes it an effective antifreeze, as it can absorb and release heat quickly to help prevent the formation of ice. As a fuel additive, propanol can improve the combustion characteristics of gasoline, making it more efficient and reducing emissions.
While propanol's low specific heat capacity is beneficial in many applications, it can also be a drawback in certain situations. For example, propanol's low heat capacity means that it can evaporate more quickly than other substances, which can make it less effective as a heat transfer medium in certain applications. It can also be more prone to overheating when used in high-temperature environments.
In conclusion, the specific heat capacity of propanol is an important property that affects its use and behavior. Its low specific heat capacity makes it an effective solvent and antifreeze, but it can also be a drawback in certain situations. Understanding the specific heat capacity of propanol and how it relates to its properties and uses is important for effectively utilizing this versatile substance.