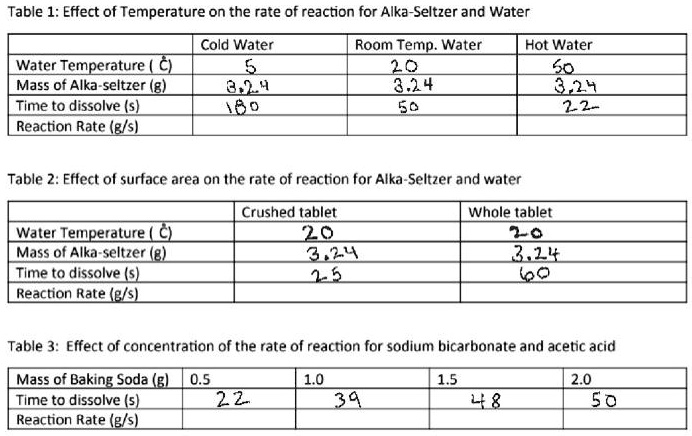
Alka-Seltzer is a popular over-the-counter medication that is used to treat symptoms of indigestion, heartburn, and upset stomach. It is composed of three active ingredients: aspirin, citric acid, and baking soda (sodium bicarbonate). When Alka-Seltzer tablets are dropped into water, a chemical reaction occurs that releases carbon dioxide gas, creating the fizzing and bubbling that is characteristic of the product.
The reaction between the active ingredients in Alka-Seltzer and water can be represented by the following equation:
Aspirin (C9H8O4) + citric acid (C6H8O7) + sodium bicarbonate (NaHCO3) + water (H2O) → carbon dioxide gas (CO2) + sodium citrate (Na3C6H5O7) + water (H2O)
In this equation, the reactants (the substances on the left side of the arrow) are the active ingredients in Alka-Seltzer and water. The products (the substances on the right side of the arrow) are the substances that are formed as a result of the reaction.
The aspirin in Alka-Seltzer is a pain reliever that is commonly used to treat headaches, muscle aches, and other types of pain. It is a weak acid that is converted to salicylic acid (C7H6O3) when it is dissolved in water.
The citric acid in Alka-Seltzer is a weak organic acid that is commonly found in citrus fruits such as lemons and oranges. It is used in the product to help neutralize excess stomach acid and to provide a sour taste.
The sodium bicarbonate in Alka-Seltzer is a base that is commonly used to neutralize excess acid in the body. It is also used in baking as a leavening agent to help baked goods rise.
When Alka-Seltzer tablets are dropped into water, the active ingredients dissolve and the reaction between the aspirin, citric acid, and sodium bicarbonate releases carbon dioxide gas, which creates the fizzing and bubbling effect that is characteristic of the product. The sodium citrate that is produced in the reaction helps to neutralize excess acid in the stomach, helping to relieve symptoms of indigestion, heartburn, and upset stomach.
In conclusion, the chemical reaction that occurs when Alka-Seltzer is dropped into water can be represented by the equation given above. This reaction releases carbon dioxide gas, which creates the fizzing and bubbling effect, and produces sodium citrate, which helps to neutralize excess acid in the stomach.